 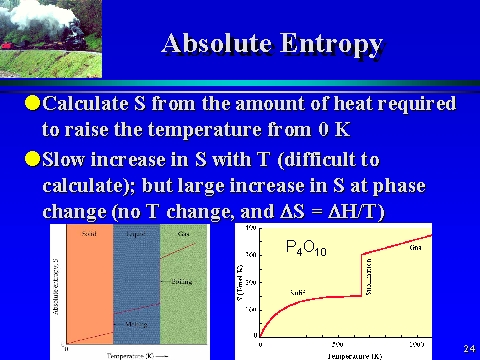
In the second expression the heat capacity C may be expressed in terms of the molar heat capacity C m (units: J K -1 mol -1), as C = nC m. In the first expression, n is the amount of gas molecules (units: mol) and R is the gas constant (units: J K -1 mol -1), which is defined in terms of Boltzmann's constant k (units: J K -1) and Avogadro's constant N A (units: mol -1) by R = N A k. Three applications of the above expressions are as follows: In practice, the external pressure or temperature needs to be infinitesimally different from that of the system to ensure the appropriate direction of change. In reversible heating, the temperature of the external heater is matched to the changing temperature of the system at all stages of the heating: there is thermal equilibrium throughout. In a reversible expansion, the external pressure is matched to the changing pressure of the system at all stages of the expansion: there is mechanical equilibrium throughout. In a reversible process, the system and its surroundings are in equilibrium. A reversible process is one that changes direction when an external variable, such as pressure or temperature, is changed by an infinitesimal amount. 
The definition refers to the reversible transfer of energy as heat. It is commonly demonstrated that S is indeed a state function by using the Carnot cycle, as explained in textbooks. It is far from obvious that the definition in (1) implies that S is a state function, or equivalently that d S is an exact differential (one with a definite integral that is independent of the path of integration). 1Įntropy is a state function that is, it has a value that depends only upon the current state of the system and is independent of how that state was prepared. With energy (and, by implication, heat) in joules (J) and temperature in kelvins (K), the units of entropy are joules per kelvin (J K -1). Where n and m are the coefficients found in the balanced chemical equation of the reaction.Where q rev is the total energy transferred reversibly during the change of state. The entropy change of a reaction where the reactants and products are in their standard state can be determined using the following equation: (Source: UC Davis ChemWiki by University of California\CC-BY-SA-3.0) Standard Entropy Change of a Reaction, Δ S° Temperature of a Single Substance.” This is a generalized plot of entropy versus temperature for a single substance. These large increases occur due to sudden increased molecular mobility and larger available volumes associated with the phase changes.įigure 18.3 “Entropy vs. This can be seen in Figure 18.3 “Entropy vs. Temperature of a Single Substance.” Large jumps in entropy occur at the phase changes: solid to liquid and liquid to gas. The standard molar entropy of any substance increases as the temperature increases.Gases tend to have much larger standard molar enthalpies than liquids, and liquids tend to have larger values than solids, when comparing the same or similar substances. 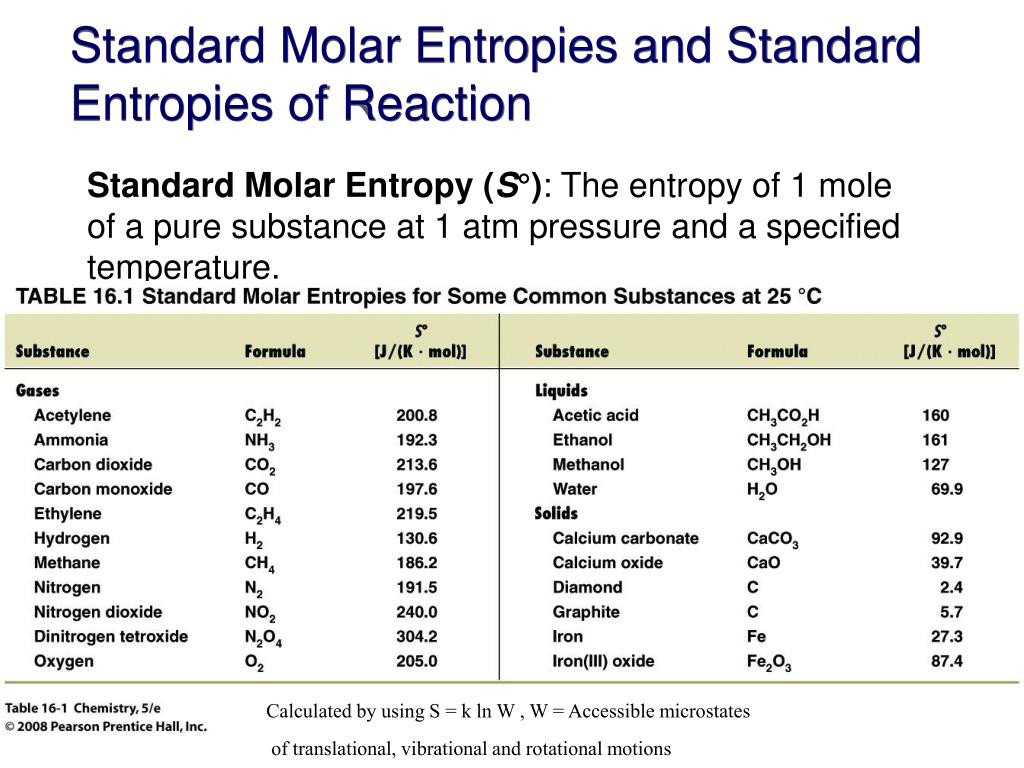
There are more possible arrangements of atoms in space for larger, more complex molecules, increasing the number of possible microstates. Larger, more complex molecules have higher standard molar enthalpy values than smaller or simpler molecules.Several trends emerge from standard molar entropy data: Table 18.1c Standard Molar Entropies of Selected Solids at 298 K Solid Table 18.1b Standard Molar Entropies of Selected Liquids at 298 K Liquid Table 18.1a Standard Molar Entropies of Selected Gases at 298 K Gas These values have been tabulated, and selected substances are listed in Table 18.1a to c “Standard Molar Entropies of Selected Substances at 298 K”. The standard molar entropy, S°, is the entropy of 1 mole of a substance in its standard state, at 1 atm of pressure. Assume the change is reversible and the temperature remains constant. 
Determine the change in entropy (in J/K) of water when 425 kJ of heat is applied to it at 50☌. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
